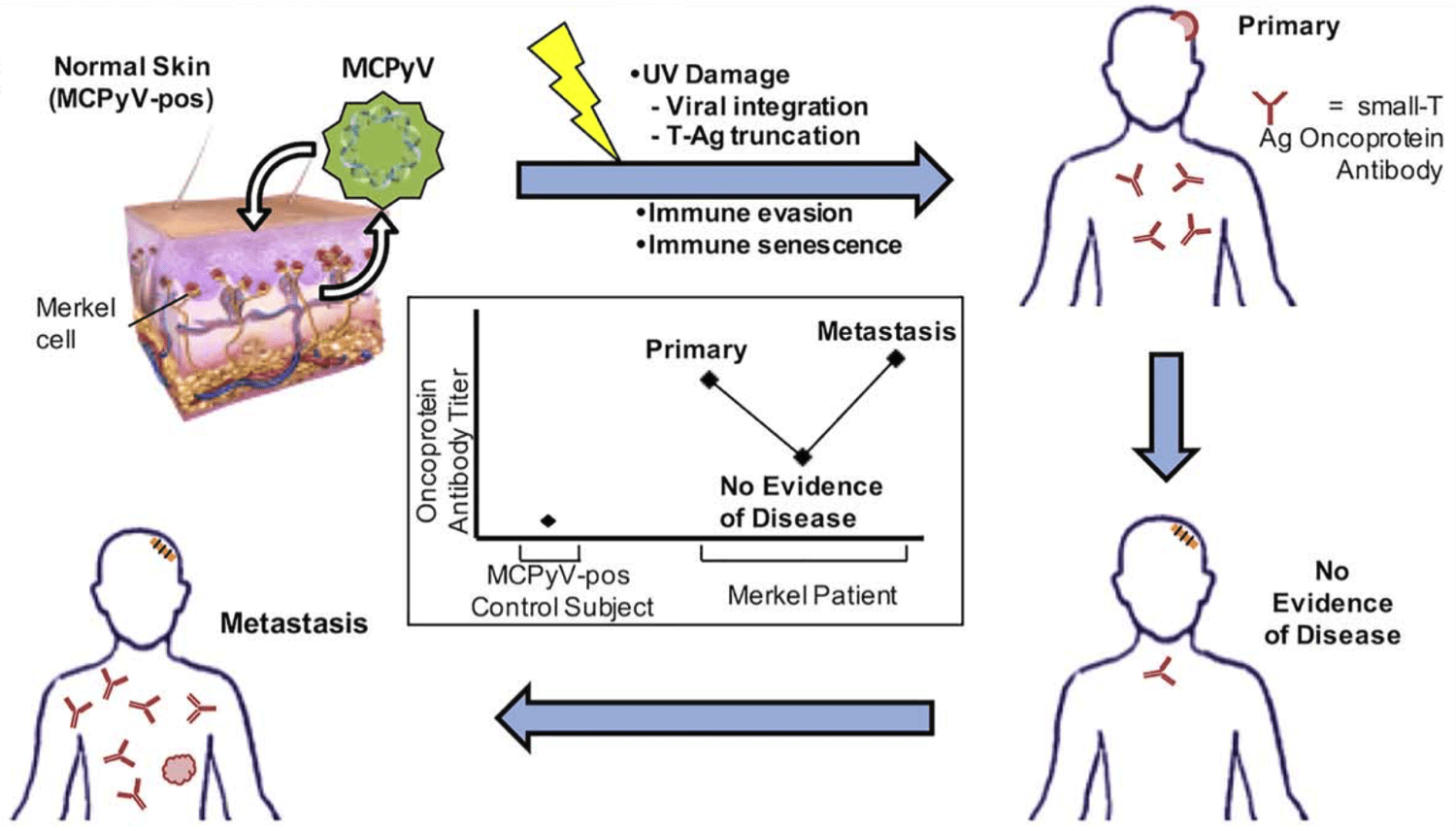
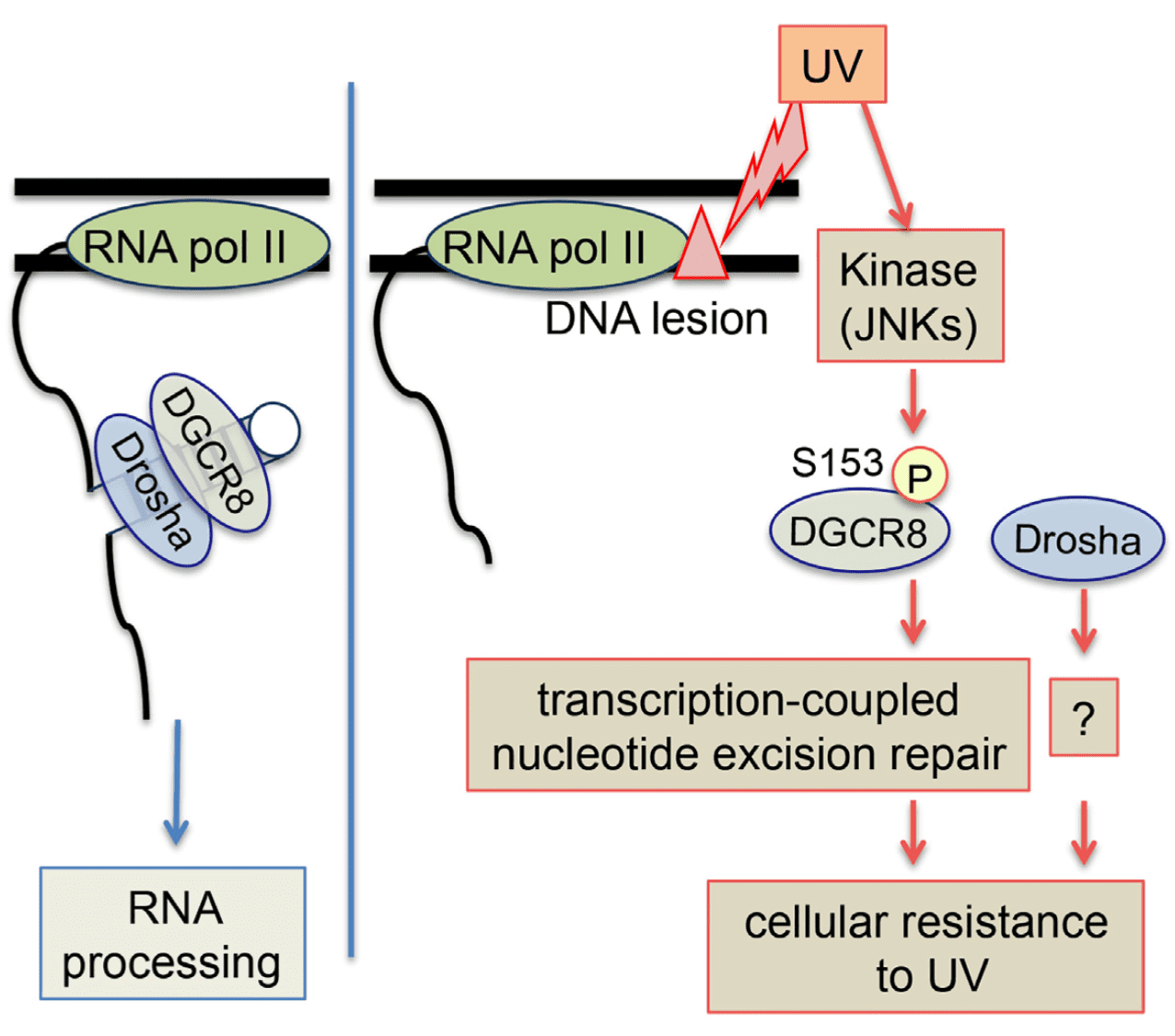
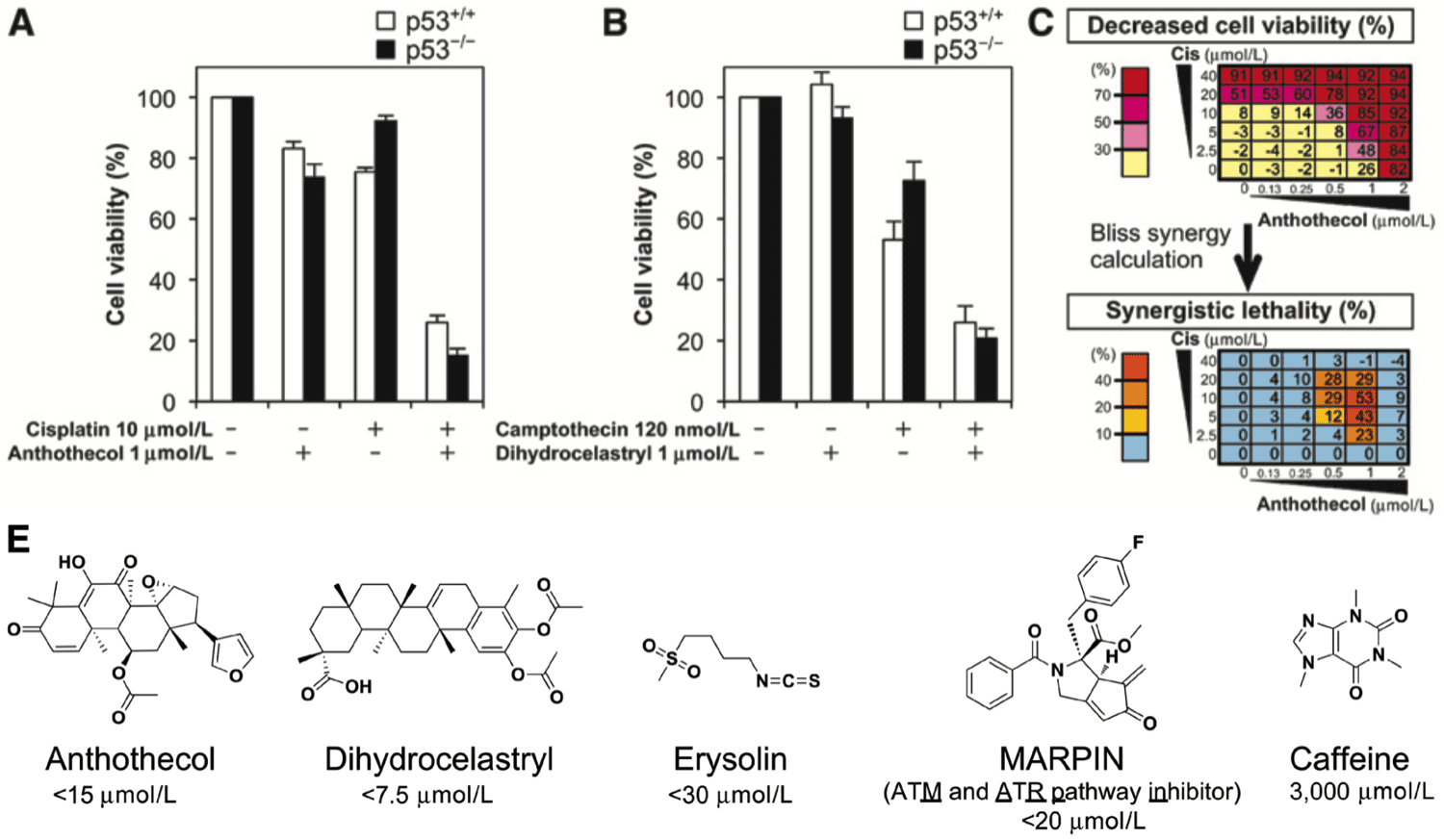
Highlights Merkel cell carcinoma (MCC) is an aggressive cutaneous malignancy with a recurrence rate of…
Abstract
The 24-residue peptide Humanin (HN), containing two Ser residues at positions 7 and 14, protects neuronal cells from insults of various Alzheimer’s disease (AD) genes and Aβ. It was not known why the rescue function of (S14G)HN is more potent than HN by two to three orders of magnitude. Investigating the possibility that the post-translational modification of Ser14 might play a role, we found that HN with D-Ser at position 14 exerts neuroprotection more potently than HN by two to three orders of magnitude, whereas D-Ser7 substitution does not affect the rescue function of HN. On the other hand, S7A substitution nullified the HN function. Multiple series of experiments indicated that Ser7 is necessary for self-dimerization of HN, which is essential for neuroprotection by this factor. These findings indicate that the rescue function of HN is quantitatively modulated by D-isomerization of Ser14 and Ser7-relevant dimerization, allowing for the construction of a very potent HN derivative that was fully neuroprotective at 10 pM against 25 μM Aβ1-43. This study provides important clues to the understanding of the neuroprotective mechanism of HN, as well as to the development of novel AD therapeutics.